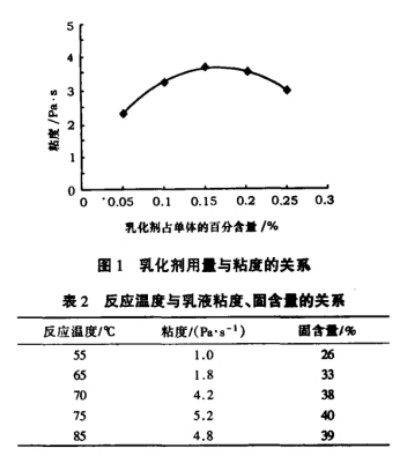
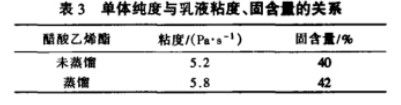
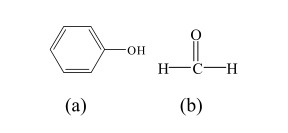
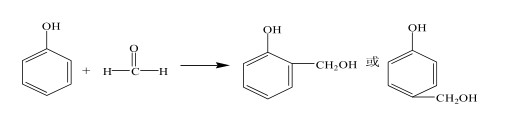
Polyvinyl alcohol (PVA) is a fundamental raw material for vinylon production and is also used in the production of adhesives, emulsifiers, and other products. In the PVA production process, solution polymerization is used to ensure a narrow degree of polymerization distribution, low branching, and good crystallinity. The VAM polymerization rate is strictly controlled at approximately 60%. Due to the control of the polymerization rate during the VAM polymerization process, approximately 40% of the Vinyl Acetate Monomer (VAM) remains unpolymerized and requires separation, recovery, and reuse. Therefore, research on VAM recovery process is a crucial component of the PVA production process. There is a polymer-monomer relationship between Ethylene Vinyl Acetate (EVA) and vinyl acetate monomer (VAM). Vinyl acetate monomer is one of the basic raw materials for making ethylene vinyl acetate polymer.
This paper uses the chemical simulation software Aspen Plus to simulate and optimize the VAM recovery process. We studied how process settings in the first, second, and third polymerization towers affect the production unit. We found the best settings to save water used for extraction and lower energy consumption. These parameters provide an important theoretical basis for the design and operation of VAM recovery.
1 Vinyl Acetate Monomer Recovery Process
1.1 Simulation Process
This process includes the first, second, and third polymerization towers in the vinyl acetate monomer recovery process. The detailed flow diagram is shown in Figure 1.
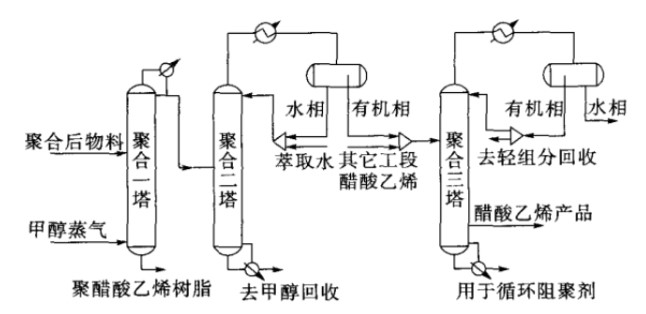
1.2 Thermodynamic Model and Module Selection
The vinyl acetate monomer recovery unit of the polyvinyl alcohol plant primarily processes a polar system consisting of vinyl acetate, methanol, water, methyl acetate, acetone, and acetaldehyde, with liquid-liquid separation between vinyl acetate and water. The main equipment in the vinyl acetate monomer recovery unit of the polyvinyl alcohol plant was simulated using Aspen Plus software. The RadFrac module was employed for the distillation tower, and the Decanter module for the phase separator.
2 Simulation Results
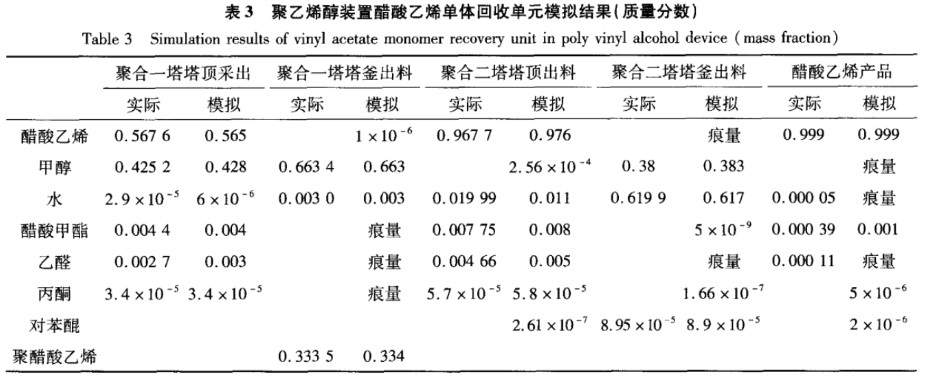
We ran a process simulation on the vinyl acetate monomer recovery unit in the polyvinyl alcohol plant. Table 3 shows a comparison of the simulation results and actual values for the main logistics. As shown in Table 3, the simulation results are in good agreement with the actual values, so this model can be used to further optimize the process parameters and process flow.
3 Process Parameter Optimization
3.1 Determination of the Amount of Stripping Methanol
Polymerization Tower 1 takes out vinyl acetate monomer (VAM) from the stream that remains after polymerization. It uses methanol vapor at the bottom for heat. The right amount of methanol is important for how well the tower works. This study looks at how different amounts of methanol affect the mass fraction of PVA at the tower's bottom and the mass fraction of VAM at the top, assuming the feed stays the same and the tower's design is constant.
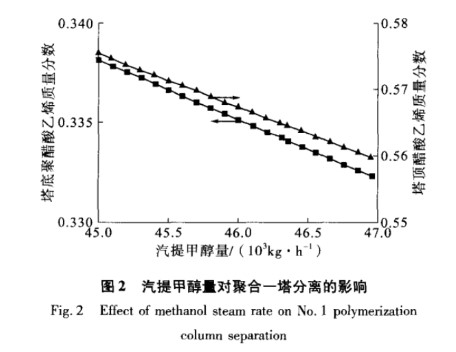
As shown in Figure 2, when the heat capacity needed for separation in Polymerization Tower 1 is satisfied, raising the stripping methanol amount lowers the PVA mass fraction at the bottom and the VAM mass fraction at the top. The stripping methanol amount has a linear relationship with the PVA mass fraction at the bottom and the VAM mass fraction at the top.
3.2 Optimization of the Feed Position in Polymerization Tower 2
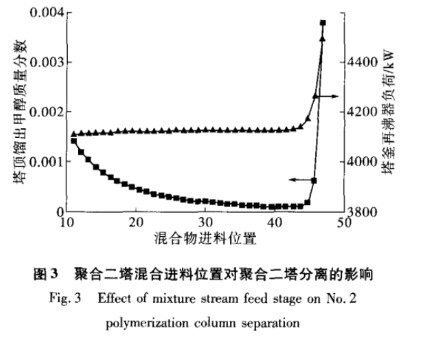
In Polymerization Tower 2, an extractive distillation tower, the locations where the solvent and feed enter greatly affect how well the separation works. This column uses extractive distillation. Based on the physical properties of the extractant and the mixed feed, the extractant should be added from the top of the column. Figure 3 shows how the mixture feed position affects the methanol mass fraction at the top and the reboiler load at the bottom, keeping other simulation settings the same.
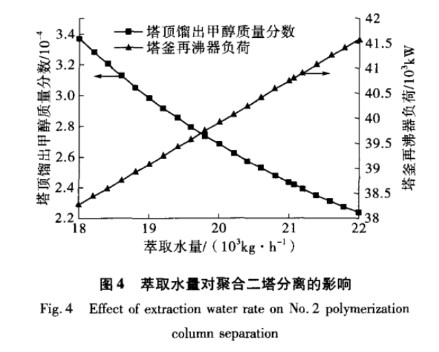
3.3 Optimizing the Extraction Water Amount in Polymerization Column 2
In Polymerization Column 2, extractive distillation is used to separate vinyl acetate and methanol azeotrope. By adding water to the top of the column, the azeotrope is disrupted, allowing for the separation of the two substances. The extract water flow rate has a big impact on how well Polymerization Column 2 separates these materials. With consistent simulation settings, I looked at how the amount of extract water affected the methanol mass fraction at the top and the reboiler load at the bottom of the column. The results are shown in Figure 4.
3.4 Optimizing the Reflux Ratio in Polymerization Column 3
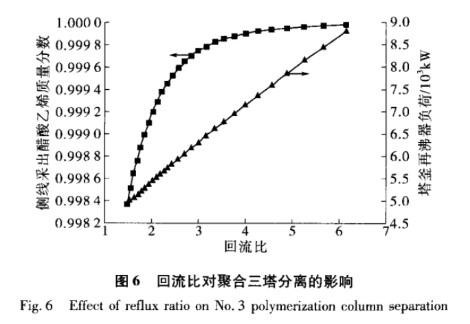
In Polymerization Column 3, the reflux ratio is important for separating vinyl acetate from lighter substances like methyl acetate and trace water. This boosts the quality of vinyl acetate obtained from the side stream. We kept the simulation settings constant and studied how the reflux ratio affects both the mass fraction of vinyl acetate from the side stream and the reboiler load. The calculation results are shown in Figure 6. Maintaining the polymerization tower's reflux ratio around 4 helps ensure the vinyl acetate from the side line meets quality standards and keeps the reboiler load low.
4. Conclusion
(1) Using AspenPlus software, a suitable thermodynamic model is selected to simulate the entire process of vinyl acetate monomer recovery of the polyvinyl alcohol plant. The simulation results are in good agreement with the actual values and can be used to guide the process design and production optimization of the plant.
(2) Based on the establishment of a correct process simulation, the influence of the process parameters of the polymerization tower 1, polymerization tower 2, and polymerization tower 3 on the plant is investigated, and the optimal process parameters are determined. When vinyl acetate meets the needed separation standards, we can save on extraction water and lower energy use.
Website: www.elephchem.com
Whatsapp: (+)86 13851435272
E-mail: admin@elephchem.com